Integrated Systems
Gravity transfer system
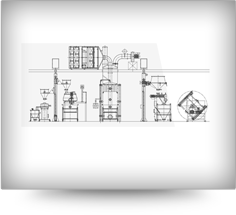 The most traditional yet the most robust system in Granulation suites which utilizes material handling
system for product transfer. The system is particularly beneficial where the ingredients are dosed
individually with good flow rate. The particles follows the integrated granulation track and hence results in
minimal human intervention with qualitative improvement.
The most traditional yet the most robust system in Granulation suites which utilizes material handling
system for product transfer. The system is particularly beneficial where the ingredients are dosed
individually with good flow rate. The particles follows the integrated granulation track and hence results in
minimal human intervention with qualitative improvement.
Multi-level transfer system
Downstream approach with smaller footprint every floor, this Suite best suited for much specialised
operation where the ordered process is the requirement for larger capacity range. The equipments are
integrated with each other through the levels with automated systems for automatic operation of the
Suite. The particles follow the integrated granulation track with minimum travel distance to achieve and
maintain the integrity of the process. Critical parameters like cleaning validation can be achieved easily.
Vacuum transfer system
Vacuum transfer suite is the Quick, direct & the most efficient system which utilizes vacuum system to
transfer product. Equipments in the suite are Integrated with each other with special fitments to achieve
and maintain the media(Vacuum) of material transfer which ensures shorter batch times &
more productivity.
Tapasya assures quality product with good documentation practices. In a cGMP environment documentation needs to meet certain requirements to ensure product quality and product safety. The cGMP regulations from FDA, PIC/S and EU all include mandatory sections on documentation.
Documents provide the information or evidence or may serve as
an official record.


 The most traditional yet the most robust system in Granulation suites which utilizes material handling
system for product transfer. The system is particularly beneficial where the ingredients are dosed
individually with good flow rate. The particles follows the integrated granulation track and hence results in
minimal human intervention with qualitative improvement.
The most traditional yet the most robust system in Granulation suites which utilizes material handling
system for product transfer. The system is particularly beneficial where the ingredients are dosed
individually with good flow rate. The particles follows the integrated granulation track and hence results in
minimal human intervention with qualitative improvement.